
Getting life-saving drugs and vaccines to underserved people in developing countries is a driving goal for many non-profit organizations. Sometimes, however, well-intentioned efforts like these can fall short of their desired impact due to poor understanding of market uncertainty and risks associated with pharmaceutical manufacturing.
Careful market analysis and optimization techniques can make the difference between a successful and stymied humanitarian project. Linksbridge SPC (a social purpose corporation) is a boutique Seattle-based consulting firm working primarily in international development. Linksbridge helps non-profits and foundations to make sound and cost-effective decisions to meet their public health objectives, increasing health impact and ‘value for money.’ “The purpose of these projects is not necessarily profitability or revenue, but the ability to meet global health goals and objectives,” says Khatuna Giorgadze, a senior associate with Linksbridge SPC who conducted the modeling and analysis for the project.
The non-profits’ objectives can range from reducing the under-five child mortality rate to combating HIV/AIDS or eradicating polio. Linksbridge’s purpose is to find the most cost-effective and impactful solution to achieve these goals. To do this, they use a range of Palisade products, including @RISK, RISKOptimizer, and PrecisionTree.
Three Stages of Analysis
A detailed analysis of market dynamics is essential to ensure access to life-saving medicines for poor countries. Linksbridge’s market analysis process typically consists of three stages: forecasting demand for a health commodity, forecasting supply, and developing market and investment strategy.
Forecasting Demand
Forecasting demand for a medical product can be quite challenging without proper tools to account for uncertainty and risk, especially as it relates to market adoption.
“It is important to differentiate here between need and demand,” Giorgadze explains. “Need is often defined as the target population that requires medical procedure, treatment, vaccination, screening etc., while demand often means that there is purchasing power behind the need. Thus, a key driver of demand variability could be countries’ fiscal readiness and/or willingness to adopt the medicine. For example, you may have 70 countries that have plans to introduce a vaccine in their immunization schedules, but there is no exact data on when they will do so. If all of them decide to adopt it in the same year, you will have very high demand—if none of them do, your demand will be zero. In many cases you cannot predict with precision product uptake or treatment adherence as well.”
With this uncertainty at play, Giorgadze opted to use @RISK to create a demand forecasting model. The model included both static inputs like population data from United Nations Population Division, country census data, and World Health Organization coverage data, as well as probability distributions for highly uncertain inputs that represent a range of possible values.
“Better demand forecasting can save lives,” says Giorgadze. Thus, using @RISK, she ran an aggregate simulation that incorporates all the uncertainties and risks. The model yielded an output which depicted demand forecast showing a possible range and likelihood of different demand outcomes.
Forecasting Supply
The next step Giorgadze took was to forecast supply of the medicine in question. This step “is even more uncertain than demand,” she says, “so we use @RISK to a great extent to assess manufacturing risks and suppliers’ plans in the market.” Key data that informs the models for this step include product development timelines, available capacity, and supplier plans to enter different markets. All of the above inputs are highly uncertain and require probability distributions to account for a variety of possible options. History of stable supply and manufacturing standards, as well as the business history and financial strength of potential suppliers also help inform analysis and assess manufacturing and regulatory risks.
Market Analysis
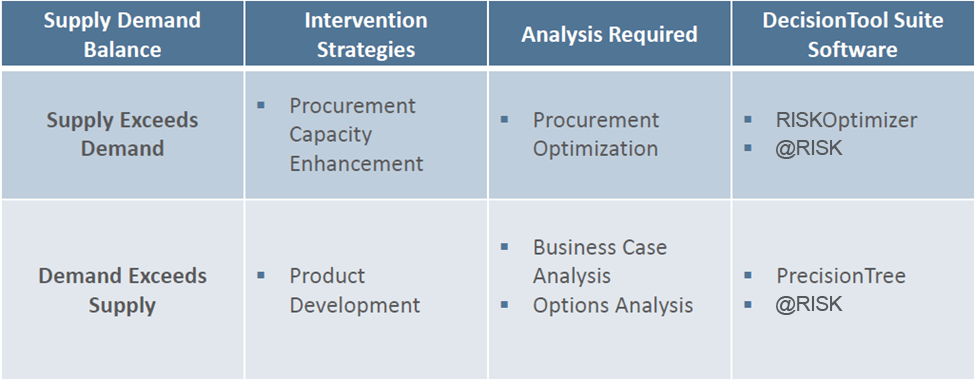
“Once we have our demand and supply picture in place, we can think through the market strategy and helpful interventions to meet the defined public health objectives,” says Giorgadze, who adds that this step relied heavily on @RISK, RISKOptimizer, and PrecisionTree. While there can be various scenarios, she focuses on the most typical cases—a situation in which supply exceeds demand, and a situation in which demand exceeds supply.
Khatuna Giorgadze
Senior Associate, Linksbridge SPC
When Supply Exceeds Demand
In the first scenario, where supply exceeds demand, the intervention strategy involves ‘procurement capacity enhancement,’ that is, structuring procurement process in such a way to:
*Obtain the lowest possible sustainable pricing *Maintain continuous and adequate supply *Secure optimal product presentations to meet the needs of patients and supply chains *Enable rapid availability of superior products to countries and patients
“Procurement is a huge market-shaping tool,” she says. “It contains a lot of opportunities to achieve value-for-money.”
In order to arrive at a procurement process that generates savings and value for money, Giorgadze built a basic optimization model and ran various procurement scenarios to compare savings. The optimization goal is to achieve the lowest total procurement costs based on different tender options and allocations decisions. Constraints in the model would be the number of suppliers in the market and what the maximum and minimum allocation would be to meet demand. Outputs for this model include optimized weighted average price (WAP), total costs, and the optimal market allocations. “This kind of simulation is extremely helpful in generating huge opportunities to save money, and are also useful for other needed health interventions,” says Giorgadze.
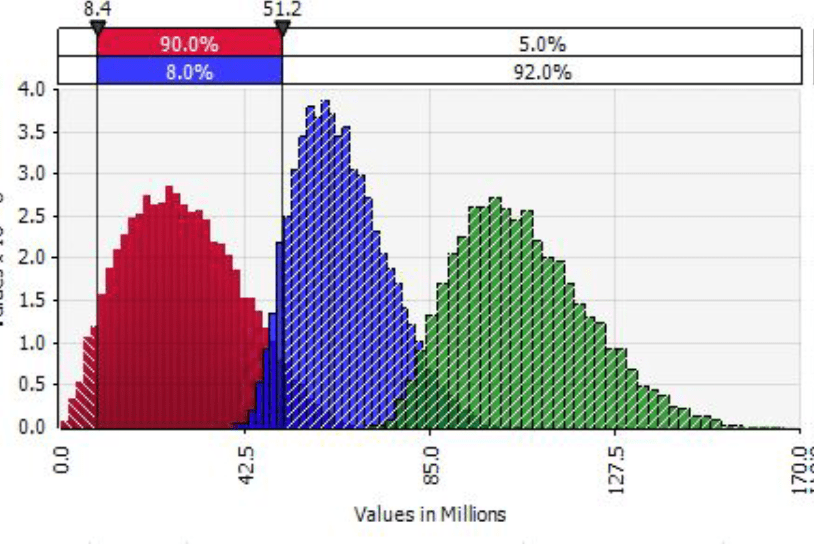 Simulation data feeds additional scenario and sensitivity analysis and generates invaluable insights on effective procurement processes.
Simulation data feeds additional scenario and sensitivity analysis and generates invaluable insights on effective procurement processes.
When Demand Exceeds Supply
In this scenario, there is a crucial need to find a supplier who can efficiently provide medicines before more lives are lost. An investment decision has to be made between two suppliers:
Option A – This supplier is a more risky choice, as uses an innovative technological platform that involves higher technical and regulatory risks, with uncertainty regarding the adoption of this product. However, it creates opportunities to fill demand, generate overall savings, and cut manufacturing costs.
Option B – This supplier makes a secure product, has robust demand and uptake, invests in capacity expansion, and has no technical or regulatory risks involved.
To make a decision between these two choices, Giorgadze built a spreadsheet model to calculate net present values (NPVs) and to define the business case for both options. “PrecisionTree helps add flexibility in defining your investment mechanism, and helps incorporate technical, regulatory and demand uptake risks to run an integrated risk analysis,” says Giorgadze.
In her example model, both investment options turned out to have positive NPVs, but have very different risk profiles. With the help of PrecisionTree, Giorgadze was able to compare different investment options potentials benefits and risks. Option A was selected as the preferred supplier for this case.
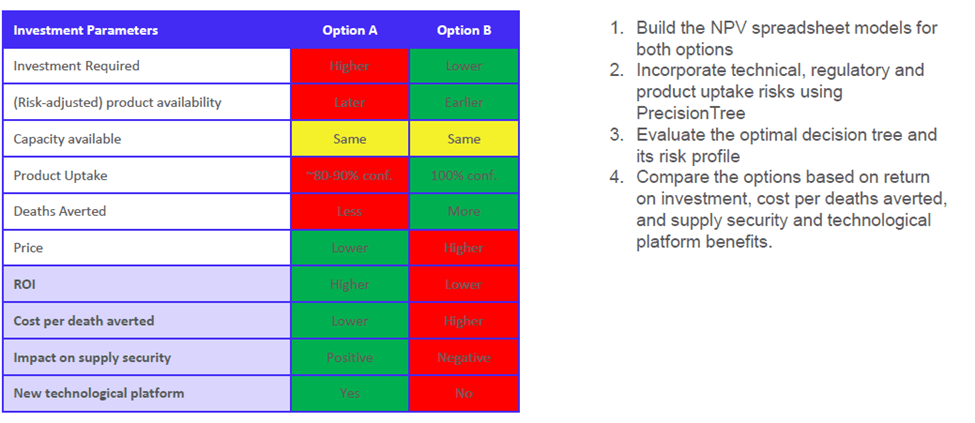 Options Analysis using PrecisionTree
Options Analysis using PrecisionTree
Options Analysis using PrecisionTree
As demonstrated by the above examples, Giorgadze was able to take complex data from multiple sources and translate it into clear and comprehensive information. This information could then be easily presented to clients looking to make sound and cost-effective investment and policy decisions to meet public health objectives. @RISK, RISKOptimizer, and PrecisionTree helped make this process all the more efficient and effective. “Palisade’s DecisionTools Suite is a very important component to our work,” says Giorgadze. “Since 2011, it has enabled us to do the required market and investment analysis and make decisions. It’s easy to use, and its compatibility with Excel makes it all the more appealing. We’re very happy with how this software has enhanced our work.”
