
AC Immune SA, a biopharmaceutical company focused on developing product candidates to treat neurodegenerative diseases, harnesses the power of Lumivero's products—specifically @RISK and PrecisionTree, to assess the value of the company’s development candidates leading to the overall enterprise value. Using @RISK, AC Immune calculates the risk adjusted net present values (rNPVs) for its preclinical and clinical drug candidates. Using PrecisionTree, the company values key decisions along the development pathway. Thanks to Lumivero, AC Immune has been able to manage risk, define prediction intervals, communicate clearly to internal stakeholders, and ask more ‘what if’ questions based off their models.
Background
AC Immune SA is a clinical-stage biopharmaceutical company leveraging their two proprietary technology platforms to discover, design and develop novel proprietary medicines and diagnostics for prevention and treatment of neurodegenerative diseases (NDD) associated with protein misfolding.
Misfolded proteins are generally recognized as the leading cause of NDD, such as Alzheimer’s disease (AD) and Parkinson’s disease (PD), with common mechanisms and drug targets, such as amyloid beta (Abeta), Tau, alpha-synuclein (a-syn) and TDP-43. AC Immune’s corporate strategy is founded upon a three-pillar approach that targets (i) AD, (ii) focused non-AD NDD including Parkinson’s disease, ALS and NeuroOrphan indications and (iii) diagnostics. They use their two unique proprietary platform technologies, SupraAntigen and Morphomer to discover, design and develop novel medicines and diagnostics to target misfolded proteins.
Using Lumivero Products
AC Immune uses @RISK to assess the Company’s enterprise value, calculating risk-adjusted net present values (rNPVs) for certain preclinical and clinical product candidates. The company then combines each respective value and determines the ultimate “Sum of the Parts” for an overall indication of the company’s price per share. The company uses this internally generated value and bridges to potential variances in their share price (Nasdaq: ACIU) or price targets published by their covering analysts.
Each product candidate that AC Immune elects to value includes many uncertain variables which impact the projected net cash flows in the development and potential commercialization period for the product candidate. The typical inputs AC Immune uses in their @RISK models include, but are not limited to:
- Clinical development timelines
- Epidemiological data
- Market share
- Estimated price per region
- Launch uptake and period
- Probability of success through development
- Discount rate
“Certain of these variables can be material and are difficult to derive a point estimate for, or can be difficult to otherwise source,” explains Julian Snow, AVP of Financial Reporting.
AVP of Financial Reporting
Typically, Snow uses a PERT distribution for his @RISK models for the risk-adjusted NPV. These inform AC Immune of the 90% prediction interval range, given the assumptions. In this example, the mean rNPV is expected to be around CHF 523m (illustrative example only).
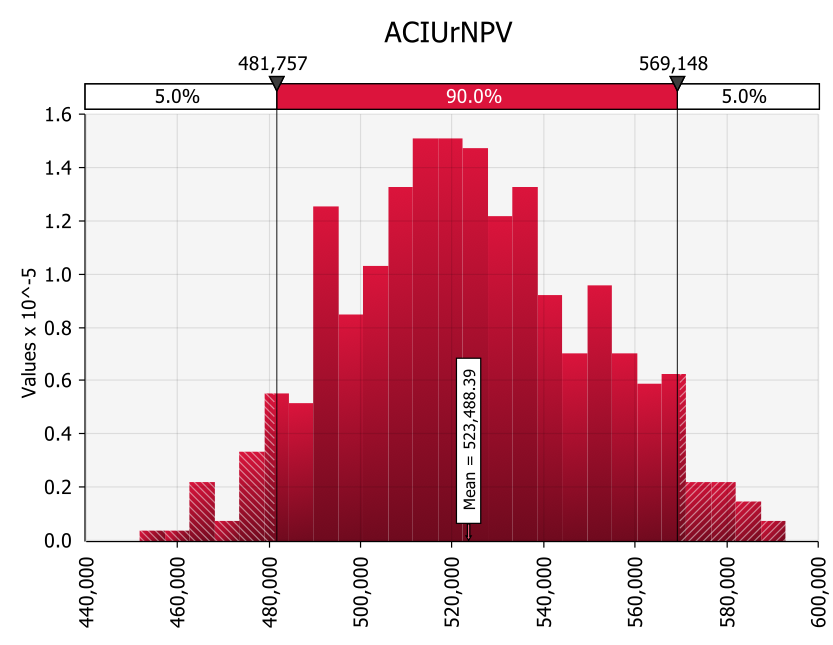
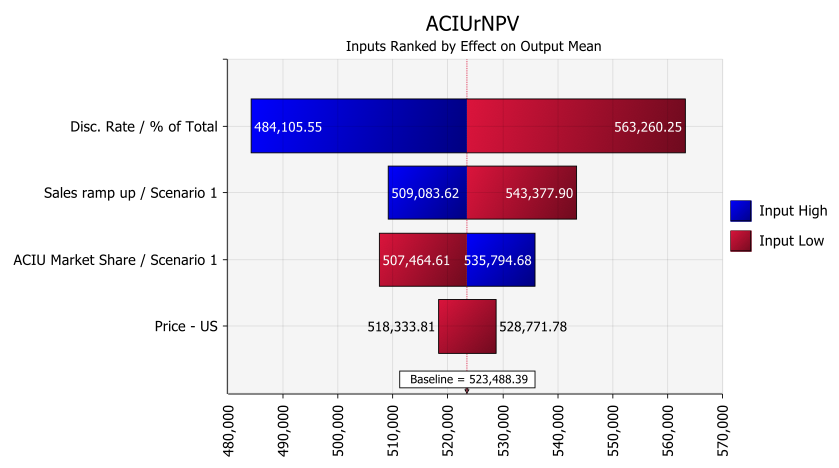
AC Immune also uses tornado charts to showcase the impact of certain assumptions on the resultant value, all other variables held constant. “The company can then decide how best to minimize the impact of certain factors via additional research into the assumption or other potential adjustments,” says Snow.
In addition to @RISK, AC Immune also uses PrecisionTree. “PrecisionTree allows us to set up dynamic decision trees linked to underlying cash flows to understand the risk/return at a specific point in time along the development timeline,” says Snow. “Additionally, it helps us weight a decision such as to partner or not partner a potential product candidate.”
Other decisions include the expansion of a program, addition of a second indication for research, or assistance in license and collaboration deal structuring. “Assessing the value of one decision or another is valuable for the company,” Snow says.
Benefits of Lumivero Products
For AC Immune, our products significantly improve the quality of the decision-making process, particularly with regard to allocation of resources and improving understanding of the magnitude of uncertainty on key assumptions.
“@RISK software allows our company to sensitize key variables using various distribution methods, as well as convey the sensitivity in impact on the ultimate risk-adjusted net present value,” says Snow. “The software also conveys results in clear output graphics for easy reporting to relevant stakeholders.”
Prior to using our products, Snow and his team relied on Excel functionalities to calculate the relevant data. “We viewed this process as static and more cumbersome to maintain,” says Snow. “Therefore, with @RISK, AC Immune was able to enhance its internal valuation and reporting capabilities.”
A Competitive Edge
According to Snow, other companies in this space do not typically leverage deterministic analysis to their valuation approaches. “Most peers use more static excel models that cannot capture or answer a more robust set of questions that arise over a long development timeline,” says Snow.
In addition, when comparing the cost-benefit of programs, assessing internal funding needs, assessing potential licensing and collaboration terms and other matters relevant to understanding the potential financial return from a product candidate, “AC Immune is able to ask and answer more questions than peers as a result of the use of the software,” Snow says.
Thanks to Lumivero's products, AC Immune has seen both tangible and intangible benefits, including:
- Managing risk via Monte Carlo simulations
- Defining prediction intervals
- Conveying information more clearly to internal stakeholders
- Allows asking of more “what if” questions based on the set up of the model
- Assess impact on rNPV when ranging discrete values
Thanks to data-driven, deterministic analysis, AC Immune’s cutting-edge drug discovery technologies are better enabled to potentially help patients around the world.












